|
FDA Voice BlogReceive this email from someone else? Subscribe now! 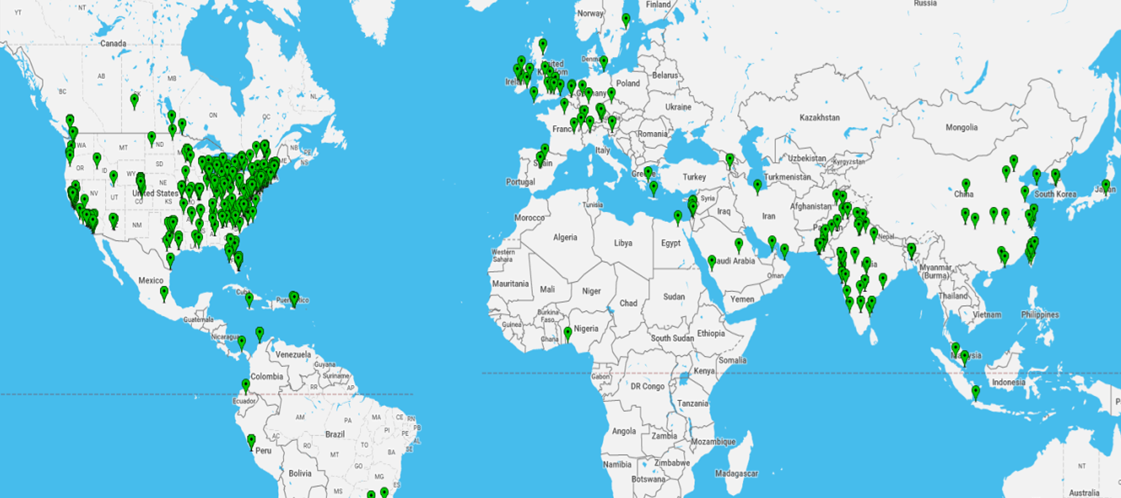
By: Brenda Stodart, Pharm.D., and Renu Lal, Pharm. D. It is well known that small business is vital to the success of the American economy. Less known, though, is how instrumental it has been to the growth and innovation in drug development. For many years FDA has been assisting small pharmaceutical companies to maximize their opportunities for success. The Generic Drug Forum on April 4-5, 2017, is one example of the work we do to support small businesses. Organized by FDA's Center for Drug Evaluation and Research Small Business and Industry Assistance (SBIA) staff, representatives from a wide range of pharmaceutical companies will gather to learn about the development, testing, review, and approval of generic drugs. CDER SBIA holds at least four meetings a year as part of a series called the Regulatory Education for Industry (REdI) conferences. REdI conferences typically attract significant international attendance (in-person or via webcast). This global reach is important, as about 80 percent of active pharmaceutical ingredients used in U.S-manufactured drugs come from more than 150 different countries. The map shows the geographic distribution of our most recent REdI conference registrants. Thirty percent of registrants were from outside the U.S., representing 55 countries worldwide. Continue reading |
We have Your e-Wallet ready. Claim in 3hrs
Hey! I saw that you haven't opened your E-wallet yet and I was wondering if... > You can check the wallet here You should really use your e-wallet because I just transferred you something special, take it via the link below: Open your E-Wallet Regards, Payton Wyman. Unsubscribe

Comments
Post a Comment